Reprinted with permission from Quanta Magazine’s Abstractions blog.
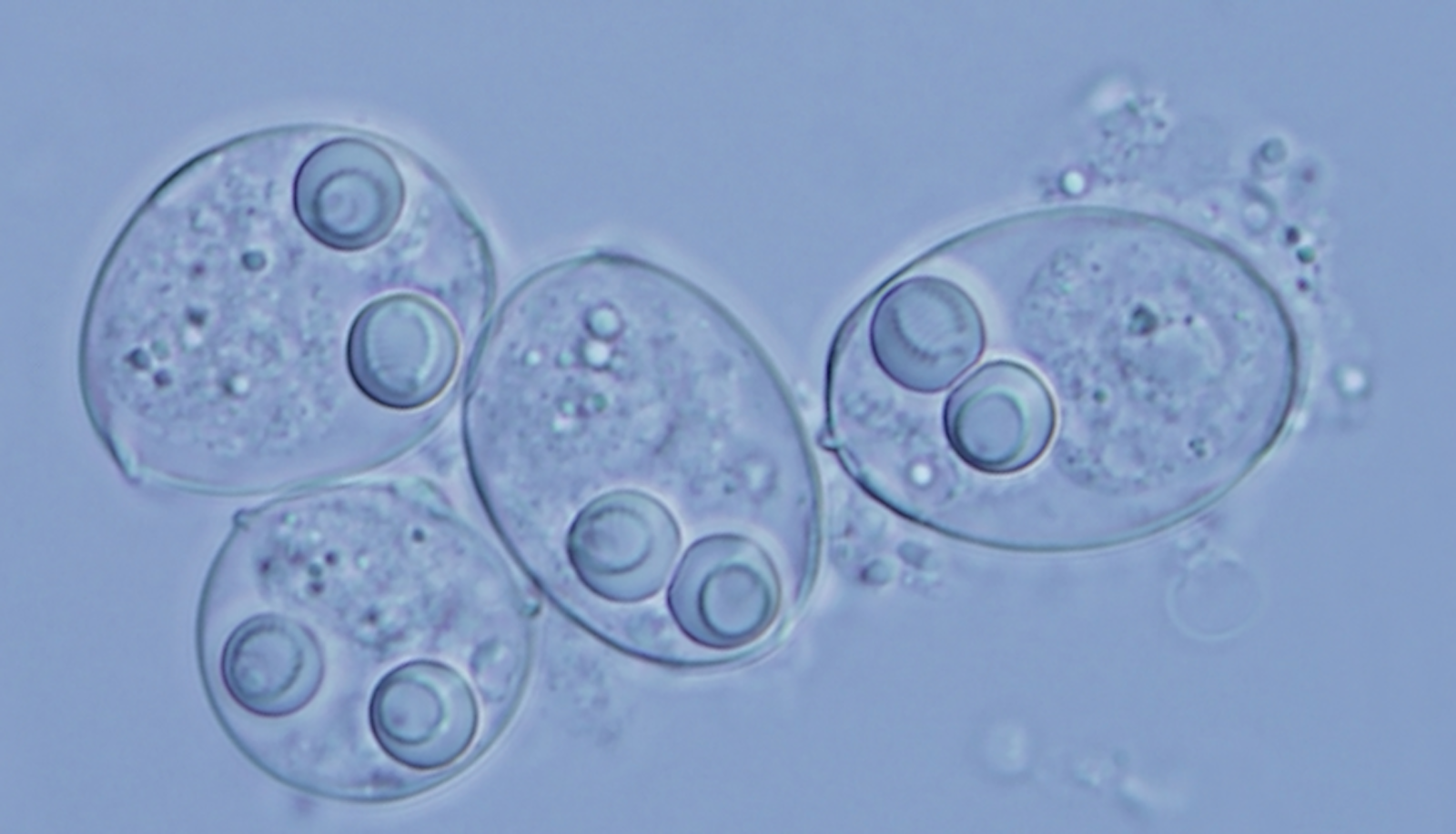
Aggressive cancers can spread so fiercely that they seem less like tissues gone wrong and more like invasive parasites looking to consume and then break free of their host. If a wild theory recently floated in Biology Direct is correct, something like that might indeed happen on rare occasions: Cancers that learn how to roam between hosts may gradually evolve into their own multicellular species. Researchers are now scrutinizing a peculiar group of marine parasites called myxosporeans to see whether they might be the first known example.
Even among microscopic parasites, myxosporeans are enigmatic. They were first discovered nearly two centuries ago, and more than 2,000 species are recognized today. Their complex life cycles make study particularly difficult: It wasn’t until the 1980s that scientists realized the ones found in fish were the same species as those found in worms, and not completely different classes of parasite. And while most parasites are content merely to snuggle into their animal host’s tissues, myxosporeans often take up residence inside a host’s own cells.
It’s not clear how or why a complex multicellular creature discarded these seemingly necessary genes along with huge chunks of its DNA.
Until fairly recently, myxosporeans were considered to be protists, offshoots of the eukaryotic line that are neither plants, animals, nor fungi. In 1995, however, Mark Siddall, then at the Virginia Institute of Marine Science, and his colleagues argued that myxosporeans are weird members of the cnidarians, the group that includes jellyfish and corals. Since then, genetic studies have bolstered that position.
But their location on the tree of life doesn’t explain how myxosporeans ended up with such strange traits. Myxosporeans boast some of the smallest known animal genomes. The genome of Kudoa iwatai, for example, is estimated to be a mere 22.5 megabases, considerably smaller than that of any other cnidarian genome. It’s less than one-twentieth the size of the genome of Polypodium hydriforme, a closely related cnidarian parasite.
Moreover, their genomes have not just been catastrophically reduced. They specifically lack certain genes thought to be essential for multicellular life. It’s not clear how or why a complex multicellular creature discarded these seemingly necessary genes along with huge chunks of its DNA.
Yet Alexander Panchin, a senior researcher at the Russian Academy of Sciences, and his colleagues have an intriguing if controversial hypothesis to explain it. Early this year, they proposed that myxosporeans initially branched off from their cnidarian kin not as independent animals, but as tumors.
Evolutionary Scandals
Panchin knows the idea of cancer-derived animals sounds far-fetched—so much so that, in the paper, he and his co-authors refer to them as Scandals (an acronym for “speciated by cancer development animals”).
At first, Scandals were just a thought experiment. While Panchin was writing about transmissible cancers, he heard his colleagues express surprise at the genes for complex tissues that were turning up in certain unusual but simple parasitic animals. Further conversations led to what Panchin calls the “fantastic” idea that such simple parasites could have cancerous origins. “So we took all the data and we proposed this hypothesis,” he said.
According to Panchin’s three-step scenario, a Scandal would start off as a cancer, but not just any cancer. It would have to be transmissible, so that it wouldn’t die when its host did. Then the cancer would have to spread to other species, and then independently evolve multicellularity. Those steps might seem to present insurmountable barriers, and yet there’s reason to believe each one could have happened.
The first step, the emergence of the transmissible cancer, is the most straightforward because we know it happens, although it is rare. Devil facial tumor disease (DFTD) has become notorious as a transmissible cancer devastating Tasmanian devils, who transmit it to one another in their bites. More common but perhaps less famous is canine transmissible venereal tumor (CTVT), a sexually transmitted disease among dogs that, according to a recent analysis by Elizabeth Murchison of the University of Cambridge and her colleagues, has been evolving as a transmissible cancer for as long as 8,500 years. (In a 2014 report, Murchison and her co-authors described CTVT as perhaps “the oldest and most widely disseminated cancer in the natural world.”)


Transmissible cancers are not confined to mammals; they have also been found in mollusks. There’s no reason to think it would be impossible for transmissible tumors to arise in a cnidarian too. Cnidarians certainly aren’t immune to cancers in general. If myxosporeans are Scandals, they most likely began as tumors of other cnidarian parasites—such as their Polypodium cousins, for instance.
Although the spread of a cancer to other species might seem unlikely, “it’s not unheard of,” said Athena Aktipis, an assistant professor at Arizona State University. Aktipis, who specializes in the evolution of cancer, points to cases such as that of a man with HIV who was discovered to be infected with tumor cells from a tapeworm. Such worm cancers have turned up repeatedly among people with compromised immune systems, and the known cases likely represent only the small minority of occurrences in which the source of a strange growth was tracked down. If this kind of species hopping happens right before our eyes, “maybe we should also consider the possibility that things that were cancer or cancerlike sometimes, in the right conditions, could become parasites on other species,” she said.
“I think that the field has been way too cautious about talking about when cancer becomes its own species, or its own kind of organism,” Aktipis said. In her view, researchers have seen too many examples of transmissible tumors like CTVT and DFTD. “It’s a parasite. It’s a parasitic organism.”
Perhaps the least likely step in the Scandal hypothesis is the one where the cancerous parasite evolves from a unicellular existence to a multicellular one with distinct hosts and stages. Myxosporeans are simple animals but truly multicellular—so if they arose from a transmissible tumor, that tumor would have had to evolve distinct cell types.
Multicellularity is thought to have evolved at least 25 times in eukaryotes, the domain of life that includes complex single-celled creatures as well as plants, animals, and fungi. In animals, though, it’s believed to have arisen just once at the very base of our lineage. Some multicellular branches of the eukaryotes have reverted to unicellularity, but no animals have been known to do so (unless, like some scientists, you consider cancer itself to be a kind of reversion). As yet, there don’t seem to be lineages of any kind in which multicellularity was gained, lost, and then gained again, in keeping with the Scandal hypothesis. “We understand that this is a very improbable scenario,” Panchin said.
But that doesn’t mean it couldn’t have happened. “I think it’s certainly possible that clusters of cancer cells that are transmissible could evolve to have something like a life cycle,” Aktipis said. “There’s nothing special about the evolutionary process that says you can only evolve a life cycle if you are a branch of the evolutionary tree that didn’t derive from [a part of] another organism.”
Following the Evidence
In the hope of finding more substantive evidence for the Scandal theory, Panchin and his team compared the genomes of a variety of simple species (most of them parasitic) with those of five myxosporeans, three single-celled creatures, and 29 other animals. They looked for hints of a cancerous past by checking for the absence of genes that are often lost when cells turn malignant. These include genes involved in apoptosis, the regulated self-sacrifice that purges abnormal cells from the body. Any organism evolving from a transmissible tumor would presumably lack such genes.
Although the scientists had expected other parasites to be the most likely Scandal candidates, only the myxosporeans had lost key tumor-suppressing genes. So they drilled deeper and found that the myxosporeans have lost so many genes related to apoptosis that they probably can’t trigger that death pathway at all. That deficiency stood out: “Even if you look at very simplified parasites which are animals, we don’t see this degree of lack of cancer-related genes,” Panchin said.
“I think that the field has been way too cautious about talking about when cancer becomes its own species, or its own kind of organism.”
Aktipis thinks that Panchin and his co-authors have presented some intriguing reasons why “we should at least consider the possibility that some of the parasitic organisms that we see today might have evolved from transmissible cancers.” But it’s not case closed, she said. “This paper is a beginning for this work, not a decisive proof of it by any means.”
Juliana Naldoni, a parasitologist and myxosporean specialist with the Federal University of São Paulo, isn’t convinced that myxosporeans are Scandals at all. “They are actually much more complex than initially thought and evolve quite intricate [and specific] mechanisms of interaction with their hosts,” she said. Some species also have complex features, such as cells organized into structures resembling muscles for movement, for example. She just doesn’t find it plausible that such complexity arose from a cancer.
Adrian Baez-Ortega, a doctoral student and bioinformatician with Murchison’s Transmissible Cancer Group at the University of Cambridge, agrees with Naldoni. “It is a thought-provoking paper, if not a very convincing one,” he wrote in an email to Quanta. He isn’t terribly impressed by the loss of apoptosis genes, for example. “In the context of such a dramatic genome reduction, the claim that the lack of genes specifically related to apoptosis points to a cancerous origin seems rather cherry-picked,” he explained.
But mostly he’s skeptical that a transmissible cancer could last long enough to evolve multicellularity. Cancer cells have incredibly unstable genomes. Although this allows them to mutate rapidly and elude their host’s defenses, Baez-Ortega pointed out that on an evolutionary timescale, “this is a very detrimental strategy. As time goes on, a good portion of a cancer’s genome becomes nonfunctional or abnormal, and this might impede not just survival, but also the development of sophisticated traits like multicellularity.” The way he sees it, “even if a transmissible cancer could have survived for millions of years, it would be much more likely to remain a unicellular parasite.”
That said, he thinks the Scandal hypothesis is worth further investigation. “There is almost nothing evolution cannot do,” he said. Rather than focusing on specific missing genes, he would like researchers to scan candidate species for the diverse genomic changes that occur in cancers, from point mutations to large-scale chromosome rearrangements. “If a cancer were to become a long-lived species, all these modifications would be preserved in its genome,” he said.
Even Panchin and his colleagues aren’t going all-in on the hypothesis that myxosporeans are Scandals. “I think that’s fair to say it’s probably not true,” he said. It’s just that, with the work they’ve done so far, they can’t rule it out. “We’ve been trying to refute it with the means that we have.”
He added, “We are going to try to falsify the hypothesis through looking at the Malacosporea genome.” Malacosporeans are cnidarian parasites and the closest known relatives of myxosporeans, but they are so much more complex that they are clearly not cancer derived. If they, too, turn out to lack apoptosis genes, it would suggest that the myxosporean loss doesn’t stem from a cancerous past.
Even if, in the end, the data suggest myxosporeans aren’t evolved cancers, Panchin noted that Scandals could still be out there waiting to be discovered. “We are hoping that maybe some zoologists who have been investigating some other peculiar kind of animal at some point will say, ‘Probably those guys are wrong about Myxosporea, but this [animal], he’s obviously a cancer.’”
Christie Wilcox is an award-winning science writer who currently edits and writes scripts for SciShow. Her award-winning writing has been featured in multiple anthologies, and her bylines include National Geographic, Popular Science, and The Washington Post.