When Jon was born prematurely at 26 weeks, he weighed around two pounds and had trouble breathing on his own. For two months he lived in an incubator and eventually grew into a healthy baby and toddler. At age 4, he had two epileptic seizures. About a year later his parents began to notice that Jon couldn’t remember things that happened in his daily life. He didn’t recall watching TV or what happened at school or what book he read. Jon’s IQ was normal, he could read and write, and did well at school. He could remember facts but not episodes from the past.
By the time Jon was 19, he couldn’t find his way anywhere. He didn’t remember familiar environments, or where his belongings were kept, or routes from one place to another.
The cause of these strange impairments was revealed when neuroscientists used magnetic resonance techniques to look at his brain. They discovered that his hippocampus, the bilateral brain region deep in the temporal lobe, was abnormally small, about half the size of a healthy hippocampus. It seemed that the lack of oxygen to his brain as an infant, known as hypoxia, and the subsequent convulsions, had caused severe damage to the cells in this specific structure and stunted its growth.
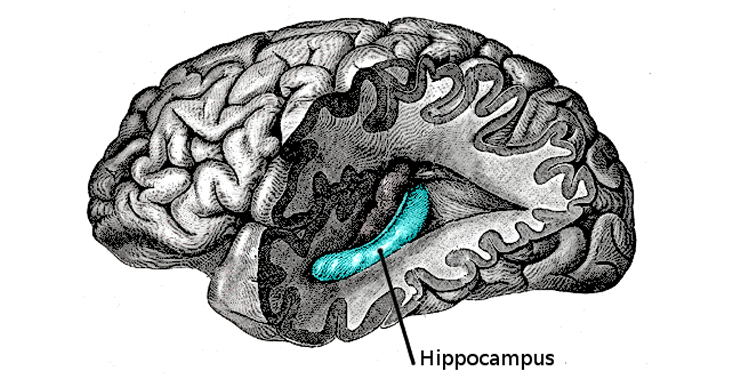
Jon, whose real name has not been made public to protect his privacy, has been the subject of research papers since the 1990s. His medical case illustrates the extraordinary function that the hippocampus plays in human life. It not only enables us to build cognitive maps of space so we can remember places and navigate, but shows that these cognitive maps are the locus of our memories of the past, what’s called episodic memory.
“The hippocampus evolved to do spatial navigation,” explains Nora S. Newcombe, a professor of psychology at Temple University. “One conjecture is that at some point in our evolutionary history, it was hijacked for the purpose of episodic memory because its neural architecture was appropriate.”
Spatial cognition and memory have deeper importance to humans beyond daily survival: They inform our sense of self. Memories of the past are like pillars of our identity; we use them to build narratives about our lives. These stories inform our actions and choices, and create a framework for imagining our possible futures.
New research is shedding light on how the hippocampus develops in infancy and childhood, a time in which circuits are maturing, and new cells are firing and encoding space to create cognitive maps. It turns out that kids’ experiences—exploring environments, navigating space, self-locomotion—can influence how the hippocampus develops.
“This is very exciting because the maturation of the brain is often considered dependent on time and a genetic program,” says Alessio Travaglia, a researcher at New York University’s Center for Neural Science. “What we’re showing is that the development of the brain is not a program, it’s about experience. So if I am a baby in New York City or in a desert or a forest, the experiences I’m facing are different.”
News of such plasticity is both fascinating and alarming. It comes at a time when pediatricians are warning that children are given less time and freedom to play, and are more sedentary than ever before.
For over a century, the absence of memory in individuals like Jon has given scientists a method for studying memory. Perhaps the most famous case of amnesia in the scientific literature is H.M., an epileptic who, in the 1950s, at the age of 27, had part of his temporal lobe removed and lost his ability to acquire and recollect episodic memories. It was H.M.’s case that led scientists to identify the hippocampus as the source of episodic memory.
Fascinatingly, each of us is like H.M. and Jon. We are amnesiacs when it comes to our first years of life. We can’t recall events before the age of 2 and our memories are typically sketchy and unreliable until the age of 6. This strange phenomenon is called infantile amnesia, followed by a period of childhood amnesia, and its presence in humans as well as other species, from rats to primates, has been a mystery for decades.
“Everyone thinks the first two years are so important. But if we can’t remember them, how are they important?” says Newcombe, Principal Investigator of the Spatial Intelligence and Learning Center at Temple University. “There are some answers but if we can’t answer it crisply, that tells us we don’t really understand anything about the brain.”
Everyone thinks the first two years are so important. But if we can’t remember them, why are they important?
Sigmund Freud, who coined the term infantile amnesia, explained it in terms of repression: The brain was hiding the desires and emotions of infancy from the adult psyche, which could be accessed through psychotherapy. Subsequent explanations for infantile amnesia sought to debunk his idea, and posited that children’s capacity for long-term memory was made possible by their acquisition of language. But other species who have also been shown to have a period of infantile amnesia never develop language, casting doubt on the idea.
In 1978, neuroscientists Lynn Nadel and John O’Keefe published a landmark book called The Hippocampus as a Cognitive Map. It presented the theory that this seahorse-shaped place in the brain is where rats, humans, and other animals create representations of their environment. These cognitive maps provide the basis for spatial memory, orientation, and navigation. Significantly, the spatial memory system stores episodes and narratives from our autobiographies. Indeed, memories of our experiences are infused with a spatio-temporal context. When we recall something that happened long ago, we engage in mental time travel and visualize the “where” and “when” of the past.
The theory was supported by O’Keefe’s earlier discovery that the rat hippocampus contained neurons that he called place cells, which fired when the animal was in a new or familiar environment. Different place cells, active in different parts of an environment, create cognitive maps. The finding would win O’Keefe a Nobel Prize in 2008, and scientists have since discovered other critical cells in the hippocampus used for spatial memory and navigation. Some of these include head-direction cells, which discharge in relation to which way our head is pointed on the horizontal plane, and grid cells, which fire as we roam an environment, building a coordinate system for navigating.
The firing of these cells is spurred by movement, exploration, and the experience of novel and familiar environments. There’s evidence that the richness and complexity of the environment influences the quantity of neurons and subsequently the volume of the hippocampus. In 1997, for instance, researchers found that mice exploring enriched environments—paper tubes, nesting material, running wheels, and rearrangeable plastic tubes—had 40,000 more neurons than a control group. These additional neurons resulted in an increase in hippocampal volume of 15 percent in the mice, and significant improvements in their performance on spatial learning tests. The researchers concluded that a combination of increased numbers of neurons, synapses, and dendrites led to the animals’ enhanced performance on the tests.

Nadel says it was during the writing of the book that he became interested in the developmental story of the hippocampus. Unlike other parts of the brain that are relatively mature at birth, the hippocampus matures at different times in different animals. “We had the theory of what the hippocampus did,” he says. “But what would it mean if the hippocampus didn’t work? The short answer is amnesia.” Nadel’s line of thought had brought him to a neurobiological explanation for infantile amnesia. Essentially, children like Jon can’t retain memories because they lack a fully developed hippocampus.
Nadel published a paper presenting this hypothesis in 1984. With co-author Stuart Zola-Morgan, he proposed that episodic memory is only possible after an organism’s brain is capable of place-learning, and that infantile amnesia is a period during which the hippocampal memory system for space hasn’t yet emerged.
Today, Nadel says this hypothesis is too simplistic in both its definition of infantile amnesia and in its description of the maturation process in the hippocampus. But development itself and its relationship to memory has become a key in issue in neuroscience in the past 30 years. Is the brain hard-wired to develop a system of spatial and episodic memory, or does it have to learn from experience?
“I think the field is still grappling with these issues, and we’re not exactly sure,” says Kate Jeffery, a behavioral neuroscientist who did her postdoctoral research with O’Keefe and studies hippocampal cells. But, she explains, the research to date suggests a fascinating process in which head-direction cells come online in the brain first, followed by place cells and then grid cells. So while components of the cognitive map are innate to the mammalian brain, it seems to undergo a period of acquiring spatial knowledge that may impact how well the hippocampus functions later on.
In 2010, two different research teams showed how this occurs in rats by putting electrodes into freely moving, pre-weaned rats to record individual neurons firing in the hippocampus. The teams, one at the Norwegian University of Science and Technology and the other from University College London, recorded hundreds of head-direction cells, place cells, and grid cells starting from day 16.
The teams discovered that all three of the cell types were present in the young rats a few days after they opened their eyes, before they began to leave the nest and explore their environment. But of these cell types, only the head-direction cells were fully mature. It took several weeks of exploring the environment until the place cells and grid cells became adult-like. From this data, the teams concluded that spatial learning continues to improve long after the components of the cognitive map are in place.
Children today spend significantly less time playing in outdoor environments than previous generations.
Combined research into primate and child behavior has given neuroscientists the greatest clues about how this same process might occur in children’s brains. The Swiss neuroscientists Pierre Lavenex and Pamela Banta Lavenex have proposed that around two years of age the CA1 region of the hippocampus, important for differentiating objects in long-term memory, begins to mature and counteract against infantile amnesia. Over the subsequent years of toddlerhood, the dentate gyrus, a remarkably plastic region of the brain that undergoes neurogenesis—the creation of new neurons—into adulthood, matures and supports the creation of new memories.
By the age of 7, there is a strong relationship between a child’s hippocampal volume and their episodic memory—the larger it is, the greater the ability to recall details of an event. That is precisely the age at which childhood amnesia seems to fully dissipate.
“The hippocampus isn’t a structure that isn’t there one day and then appears the next,” says Nadel. “But there is a gradual emergence of its functions; it’s the maturation of the network and the connections between those parts that give you long-term episodic memory.”
This past summer a group of researchers at New York University’s Center for Neural Science published findings that showed how susceptible the development of the hippocampus is to learning. The team chose two different developmental ages in infantile rats, postnatal day 17, which roughly corresponds to the age of 2 years in humans, and postnatal day 24, which roughly corresponds to between 6 and 10 years of age.
By measuring the levels of molecular markers in the hippocampus they showed how experience of the environment positively impacted the maturation of the hippocampus. Then by increasing or decreasing the level of these molecules, they manipulated the rat hippocampus to speed up memory retention or lengthen the window of infantile amnesia.
The researchers believe that infantile amnesia is due to a critical period—a window of plasticity when environmental stimulation actively shapes the brain. “Critical periods are when the system is particularly sensitive—if it doesn’t receive the right stimuli, it is stunted,” says Travaglia, an author of the study. “For humans, the assumption is that the brain needs the right stimulation in this critical period too. It’s a development window critical for learning a memory. And without that stimulation there will be cognitive and memory costs.”
In addition to the environment itself, another important source of stimulation to the hippocampus may be self-locomotion. In early 2016, Arthur Glenberg, a professor of psychology at Arizona State University, proposed a hypothesis that infantile amnesia starts to dissipate when children begin crawling and walking. Once babies begin moving through space rather than being carried passively, he and his co-author Justin Hayes proposed, the brain’s place cells and grid cells start firing and aligning themselves to the environment, encoding the spaces being explored and ultimately building the scaffolding of episodic memory.
Glenberg’s research for the past two decades has focused on the embodied theory of cognition, the idea that cognitive processes—both conscious and unconscious—are not disconnected from the body, as suggested by the philosopher René Descartes. Instead, the fact that we have legs, arms, eyes, ears, a motor system, and an emotional system underlie both our experiences of the world and our thinking. “It makes no sense for a faculty like cognition to have evolved without consideration of the body,” says Glenberg. “We’re not computers, we’re biological systems. We’re not programmed, we’re evolved. We should consider human cognition as flowing from the cognition of other animals.”

The notion that embodiment could contribute to solving the puzzle of infantile amnesia came to Glenberg spontaneously during a conference on childhood development. And there is intriguing evidence to supports his hypothesis. In 2007, a group of researchers in England found that the onset of crawling in 9-month-old babies was associated with a dramatic cognitive leap: more flexible and sophisticated capacity for memory retrieval. Researchers have also shown in mice that exercise and self-locomotion improved spatial learning and increased neurogenesis.
The age of self-locomotion seems to matter less than the degree of exploration children engage in; Dutch researchers found in 2014 that by the age of 4, children with a greater degree of exploration throughout childhood had a higher capacity for spatial memory and a positive correlation to fluid intelligence—solving problems, identifying patterns, and logic.
What Glenberg’s idea doesn’t fully explain is why there is such a large gap between the start of self-locomotion in the first year of human life and the reliable retention of memories around age 6. He proffered that a child needs an abundance of experience exploring space and building complex cognitive maps to develop a fully functioning hippocampal memory system anywhere near the sophistication of an adult’s.
“Your 10-month-old knows his way around the apartment but would not have much luck getting from the apartment to the park,” says Glenberg. “It takes an awful lot of experience walking around to develop this complex set of cells that are related enough they can serve as a good substrate for memory.”
We’re not computers, we’re biological systems. We’re not programmed, we’re evolved.
Newcombe, whose talk at a conference inspired Glenberg’s hypothesis, says that while speculative, Glenberg’s idea pushes the science in the right direction. For her, one of the most fascinating implications of hippocampal plasticity is how this understanding might improve medical treatment for children with disabilities that severely limit their mobility. Could giving a child an apparatus for self-locomotion during the critical period result in positive cognitive skills later on? One 2012 study showed how infants with severe motor impairment who were trained to use custom-made carts to move scored higher on cognitive and language tests than a control group. A 7-month-old child with spina bifida increased his cognition and language skills in the study at a rate greater than his chronological age.
Children today spend significantly less time playing in unstructured activities and outdoor environments than previous generations. One study found that between 1981 and 1997, free playtime decreased by almost 25 percent; another, focused on preschool-age children in Seattle, found they were sedentary for 70 percent of their day. So while the American Academy of Pediatrics recommends at least two hours of physical activity a day, most children engage in significantly less.
The new insights into the relationship between hippocampal development, infantile amnesia, and spatial cognition show that, in addition to combating problems such as obesity and ADHD, children need opportunities to explore and build cognitive maps because their cognitive health—the part of our brain intricately responsible for our very sense of self through time—depends on it. An abundance of data shows that decreased hippocampal volume is associated with addiction, PTSD, schizophrenia, and Alzheimer’s disease.
There is also alluring evidence that intelligence itself is intertwined with the brain’s spatial cognition aptitude. In September, Nature reported on a 45-year study of 5,000 “mathematically precocious youth[s],” and found a correlation between the number of their patents and peer-reviewed journal articles, and their scores on spatial-ability tests. “I think it may be the largest unknown, untapped source of human potential,” David Lubinski, one of the study’s directors, told Nature.
More and more, it seems that infantile and childhood amnesia are periods in which our brains are preparing to lay the foundation for learning through experience. While we can’t remember these earliest experiences, they shape us as human beings. “This is a part of a big enterprise of understanding the human mind and brain and its development,” says Newcombe. “It has vast implications.”
M.R. O’Connor is the author of Resurrection Science: Conservation, De-Extinction and the Precarious Future of Wild Things. She is currently writing a book about navigation, space, and memory.
Additional Reading
Vargha-Khadem, F., et al. Differential effects of early hippocampal pathology on episodic and semantic memory. Science 277, 376-380 (1997).
Tsien, R.Y. Very long-term memories may be stored in the pattern of holes in the perineuronal net. Proceedings of the National Academy of Sciences of the United States of America 110, 12456-12461 (2013).
Freed, D.M., Corkin, S., & Cohen, N.J. Forgetting in H.M.: A Second Look. Neuropsychologia 25, 461-471 (1987).
O’Keefe, J. & Nadel, L. The Hippocampus as a Cognitive Map Oxford, Clarendon Press (1978).
O’Keefe, J. Place units in the hippocampus of the freely moving rat. Experimental Neurology 51, 78-109 (1976).
Kempermann, G., Kuhn, H.G., & Gage, F.H. More hippocampal neurons in adult mice living in an enriched evironment. Nature 386, 493−495 (1997).
Nadel, L. & Zola-Morgan, S. Infantile Amnesia. In Moscovitch, M. (Ed.) Infant Memory Springer US (1984).
Palmer, L. & Lynch, G. A Kantian view of space. Science 328, 1487-1488 (2010).
Lavenex, P. & Banta Lavenex P. Building hippocampal circuits to learn and remember: Insights into the development of human memory. Behavioural Brain Research 254, 8-21 (2013).
Travaglia, A., et al. Infantile amnesia reflects a developmental critical period for hippocampal learning. Nature Neuroscience 19, 1225-1233 (2016).
Glenberg, A.M. & Hayes, J. Contribution of embodiment to solving the riddle of infantile amnesia. Frontiers in Psychology 7 (2016).
Herbert, J., Gross, J., & Hayne, H. Crawling is associated with more flexible memory retrieval by 9-month-old infants. Developmental Science 10, 183-189 (2007).
Oudgenoeg-Paz, O., Paul Leseman, P., & Volman, M.C.J.M. Can infant self-locomotion and spatial exploration predict spatial memory at school age? European Journal of Developmental Psychology 11, 36-48 (2014).
Jones, M.A., McEwen, I.R., & Neas, B.R. Effects of power wheelchairs on the development and function of young children with severe motor impairments. Pediatric Physical Therapy: the Official Publication of the Section on Pediatrics of the American Physical Therapy Association 24, 131-140 (2012).
Clynes, T. How to raise a genius: Lessons from a 45-year study of super-smart children. Nature London. 537, 152-155 (2016).
Lead collage credit: Zurijeta / Shutterstock


























