There were no eyes to see it, but the sun shone more dimly in the sky, casting its languid rays on the ground below. A thick methane atmosphere enshrouded the planet. The sea gleamed a metallic green, and where barren rock touched the water, minerals oozed out. Estuaries, creeks, and mud flats teemed with microscopic life—single-celled bacteria and archaea. This wasn’t life at its emergence on Earth. This was life on Earth at adolescence, age 1 billion.
Life thrived on the edges, where water met air and air met rock. These organisms carved out a living with metabolisms that drew energy from minerals and gases.1 But life was a slog. Sulfur and nitrogen, the elements that provided the highest energy currency at the time, were hard to come by, released slowly by weathering rocks. Hydrogen gas, another prized commodity, was voraciously consumed and turned to more methane.
Then along came a rebel, a form of bacteria that discovered a way to live on water and sunlight alone.
Outwardly, little distinguished the mutineer from other single-celled microbes of its time, other than its blue-green color, which eventually gave it its name, cyanobacteria. But on the inside, from the abundant fuels of sunlight and water, cyanobacteria generated more energy than any lifeform before. This new chemical process unleashed a gas that would forever change the trajectory of life on Earth: oxygen.
Tiny cells, preserved for 14 percent of the entire universe’s lifespan, have been waiting.
Today, oxygen feels synonymous with living, but to most organisms inhabiting adolescent Earth, it was lethal. Oxygen is highly reactive—it strips electrons off metals and possessively attaches itself to elements like nitrogen and sulfur, breaking the otherwise stable molecules most microbes had relied on for survival. Inadvertently, the new arrival was making the little food available to microbes inedible. When not reacting with other elements, O2 is so rapacious it reacts even with itself, releasing free radicals. These fragments of molecules are so unstable they will attack most chemical bonds indiscriminately, wreaking biological havoc. Exposed to high doses of such radicals, all living cells quickly corrode. It’s a wonder cyanobacteria survived their own gas. Essentially, they were setting themselves on fire.
The first cyanobacteria might have experimented with oxygen-producing energy only when they ran out of other fuel—a last resort to be used in times of desperation until they gradually evolved the ability to detoxify the oxygen. With Earth’s abundance of water and light, cyanobacteria had happened upon an untapped font of energy. Once they landed on a way to oxidize water without self-destruction, a revolution was afoot.
That revolution was photosynthesis, harnessing power from light (“phos” in ancient Greek). Cyanobacteria gathered the sun’s rays and directed their force to split water particles. Breaking water unleashes a powerful torrent of energy, which the cyanobacteria converted into energy currency, a sugar to store and feast on when needed. The possibilities photosynthesis offered were too numerous compared to the setbacks; there was no putting the genie back in the bottle.
The innovation of photosynthesis also opened the door for other microbes to profit from the energy that cyanobacteria captured from the sun. Without this incendiary reaction, there never would have been enough energy in the food web to produce more complex life, to produce moss or millipedes or house cats or us. And their descendants—modern cyanobacteria, as well as every light-hacking organism on the planet—continue to thrive today, and produce the oxygen we breathe.
Piecing together a story of organisms that lived 3 billion years ago—that were too small for our eyes to see and that lived on such a radically different world—itself feels a bit like mythmaking. We barely know a fraction of the microbes currently alive on Earth. How can we dare to concoct an origin story for one that lived so long ago?
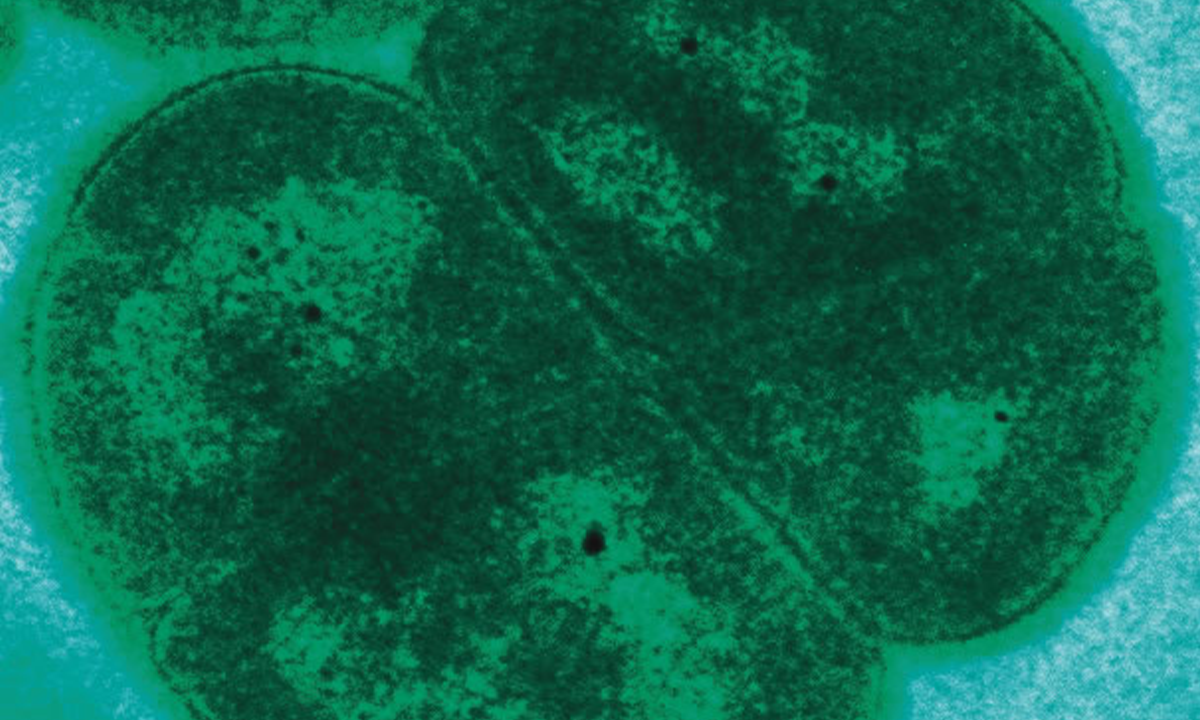
For centuries, the best and most direct evidence we have had for long-gone organisms has been fossils. And new technology has shrunk down this hunt from giant dinosaur femurs to miniscule microfossils.2 In her lab at the University of Liège in Belgium, biologist Emmanuelle Javaux uses electron microscopes—and even chemical-tracing—to search for the ancient and seemingly unseeable. The oldest cyanobacteria that have been identified in the fossil record date to the Proterozoic, a period some 1.9 billion years ago. The discovery boggles the mind: tiny cells, just 10 microns long, preserved for 14 percent of the entire universe’s lifespan. They have been waiting, in a layer at the top of a microbial mat that had turned into dense crystalline rock. It was, unmistakably, a cyanobacterium. “And the way they divide is very similar to some modern cyanobacteria,” Javaux says.
In a more recent find, Javaux has dissected cyanobacterial microfossils to reveal intricate structures within their cells—small membranes that present the first “direct evidence of a cell performing oxygenic photosynthesis,” she says.3 The tiny cells in question are 1.75 billion years old. Additional chemical analysis can reveal clues about early cyanobacteria’s metabolism and biology. But, for Javaux, “I am always amazed to find such old cells and look at them under the microscope … they are treasures.” Even these incredibly ancient microfossils, however, are roughly 1 billion years too young to reconstruct a proper origin story of the intrepid inventors of photosynthesis.4
In the absence of ancestral fossils, other scientists are scrutinizing the genomes of microbes alive today, the living descendants. “Because we’re using genes and phylogenies, we can work backward in time earlier than the fossil record,” says Greg Fournier, professor of geobiology at MIT. Identifying pieces of microbial genomes that are the oldest becomes scientists’ first step to building a partial profile of the original rebel.
But tracing the evolutionary path backward using only modern cyanobacteria is also difficult. “We don’t have direct ancestors,” Fournier says. “We have this big, long branch in our tree that separates cyanobacteria from everything else. Along that branch, all of the evolution of oxygenic photosynthesis must have occurred,” but the stepping-stones are unknown. “Maybe they’re out there … but we haven’t found them yet,” Fournier says. “It’s a little bit like trying to understand something about human evolution without any knowledge of the great apes,” adds Woodward Fischer, professor of geobiology at Caltech.
Another intriguing way to decipher the history of these planet-changers is to study the environmental signature of their exhales. Because of oxygen’s reactive properties, particularly the rusting effect it has on iron, oxygen emissions on a large scale leave a discernible trace in the rock record. A major geological turning point in our planet’s history is the “Great Oxidation Event”—or GOE for short—dated to 2.4 billion years ago. It is then that oxygen gas for the first time reached a level of 1 to 5 percent of the atmosphere’s composition (still just a gasp from today’s 21 percent). Given that much of the initial gas produced would have reacted with other chemicals and been immediately sucked up, scientists conjecture that only sustained cyanobacterial effort could have led to such a dramatic buildup in the planet’s atmosphere.
Today, oxygen feels synonymous with living, but to most organisms then, it was lethal.
But the first “whiffs of oxygen,” prior to the GOE, remain difficult to pinpoint.5 To search for traces, some scientists are plumbing ancient rocks. Using new methods, one team of scientists, including Timothy Lyons, a professor of biogeochemistry at the University of California, Riverside, asserted that they had detected minute “pulses of oxygen“ in the atmosphere, in localized environs, at least some 50 million years prior to the kickoff of the GOE. It is possible that these are either the vestiges of cyanobacterial experimentation, as described by Fournier, or these emissions may have non-biological sources. There are certain geological scenarios where minerals are deformed under mechanical stress and forced to react with water, releasing oxygen.6
These might also be parallel narratives converging on a single story: Cyanobacteria first evolved 3 billion years ago, but it took them—and likely an assist from geological processes—another 600 million years to oxidize the planet. “You can divide Earth history in about half,” Fischer says, “there’s a half without oxygen and a half with oxygen,” before and after the GOE—before and after cyanobacteria gained steam with their revolution.
That cyanobacteria prevailed is astounding not just on the level of each individual organism surviving the toxic assault of oxygen, but also given the catastrophic impacts accumulated oxygen had on the climate. Some scientists have dubbed the GOE the “Great Oxygen Catastrophe.” Not only did it poison the air, but it also stripped the atmosphere of its methane blanket. Without a warming mechanism, the globe spiraled into a period of deep glaciation. Temperatures plummeted so low, a sheet of ice stretching from the poles to the equator turned the Earth into a giant “snowball.” This ice age lasted 85 million years. The survivors eked out an existence perhaps reminiscent of the microbes that live in today’s polar ice.
The relentless oxygen levels, a lack of nutrients, and the cold temperatures would have turned most of the places where microbes had thrived for 1 billion years entirely uninhabitable. All from a single spark, a microscopic chemical treachery.
With oxygen ascendent, all of Earth’s previous life forms—obligate anaerobes (bacteria and archaea that cannot tolerate even miniscule quantities of O2)—were forced into refuge in environments free of oxygen. They persist in such refugia today, in stagnant pools, mud pots—like the bubbling acid hot springs at Yellowstone National Park—or near hydrothermal vents deep in the ocean, pockets of the planet that retain echoes of what most of it once was like.
Temperatures plummeted so low it turned the Earth into a giant “snowball.”
Other organisms got creative, looping in the newly abundant compound for their own benefit. One spinoff was respiration, the metabolic process by which most cells that live exposed to oxygen break sugars to release energy. Respiration is an ancient metabolic invention and likely evolved just after photosynthesis. But respiration can’t outpace photosynthesis; to remain a dominant force in the atmosphere, O2 must be continually produced.
The single, rebellious invention of exploding water to release energy has not been repeated in the 3 billion years of continuous innovation in evolution, since cyanobacteria first emerged. Instead, it has passed along the tree of life like a torch of Promethean flame.
The vegetation that populates our Earth, coloring our landscapes green, relies on this ancient alchemy. Instead of inventing their own version, roughly 1.5 billion years ago a bacterium engulfed a cyanobacterium and incorporated its metabolisms whole into its own cellular blueprints—giving rise to an algal cell. This next-level innovation, the enslavement of cyanobacteria, was so profound that oxygen concentrations rose exponentially as algae greened the ocean. It is estimated that today algae produce roughly half of the oxygen on the planet. Fast-forward another billion years, and a portion of these algae gradually evolved into land plants, passing the knowledge of cyanobacterial photosynthesis—nested within its cells—upward along the tree of life.
The cyanobacteria’s invention has both the simplicity and depth of a creation story. On the surface, all it brings is the union of sunlight and water to create fuel—and release oxygen. But scientists are still puzzling over the inner workings of today’s cyanobacteria. “We still don’t know all the tricks that allow them to do this so easily,” Fischer says. “You know, the best, fanciest chemists in the building across the quad from me, right?” he exclaimed excitedly during our conversation, pointing outside his office window in Caltech, “They would just love to be able to do chemistry so elegantly, in the way that it takes place in cyanobacteria, algae, and plants.”
A single organism can, unwittingly, change the atmosphere—and, with it, the entire planet’s trajectory. And although it is difficult to conjure, as we live through a climate catastrophe of our own making driven by high levels of CO2, in the constant, long fluxes of time, there might also be, “a future Earth—millions and billions of years down the line—where CO2 might be so low that it becomes limiting for oxygenic photosynthesis,” Lyons suggests. As our aging sun burns even brighter then, it might snuff all CO2 out of the air, pushing cyanobacteria and the other photosynthesizers to their twilight. “We’re not going to be around to see it—nothing like us is going to be around to see it,” Lyons says, but “the world may return to an anaerobic planet. We may return whence we came.” And the stage will be set for a new rebel. ![]()
References
1. Canfield, D.E., Rosing, M.T., & Bjerrum, C. Early anaerobic metabolisms. Philosophical Transactions of the Royal Society B 361, 1819-1836 (2006).
2. Demoulin, C.F., et al. Cyanobacteria evolution: Insight from the fossil record. Free Radical Biology and Medicine 140, 206-223 (2019).
3. Demoulin, C.F., Lara, Y.J., Lambion, A., & Javaux, E.J. Oldest thylakoids in fossil cells directly evidence oxygenic photosynthesis. Nature 625, 529-534 (2024).
4. Fournier, G.P., et al. The Archean origin of oxygenic photosynthesis and extant cyanobacterial lineages. Proceedings of the Royal Society B 288, 20210675 (2021).
5. Anbar, A.D., et al. A whiff of oxygen before the Great Oxidation Event? Science 317, 1903-1906 (2007).
6. He, H., et al. A mineral-based origin of Earth’s initial hydrogen peroxide and molecular oxygen. Proceedings of the National Academy of Sciences 120, e2221984120 (2023).