The Nobel Prize in Chemistry went to three scientists who harnessed the power of evolution and accelerated it in the laboratory to produce novel, beneficial enzymes used in pharmaceuticals, renewable energy, industrial chemistry, and many other fields. Frances H. Arnold, a professor of chemical engineering at the California Institute of Technology, received half of this year’s prize for being the first to artificially direct the evolution of enzymes; in the process, she became only the fifth woman to receive the chemistry Nobel. George P. Smith, a professor emeritus of biological sciences at the University of Missouri, and Gregory P. Winter, a research leader emeritus at MRC Laboratory of Molecular Biology, shared the other half of the prize for developing a technique called phage display, which uses directed evolution to produce pharmaceutically useful enzymes.
“For thousands of years, we humans have used selective breeding to create animals and plants with properties that have been useful for us. This year’s Nobel laureates have taken the next step,” said Claes Gustafsson, chair of the Nobel chemistry selection committee and a biochemist at the University of Gothenburg, in his remarks at the announcement. “In doing so, they have been able to make evolution many thousand times faster, and they have also been able to direct evolution to create proteins with new and useful properties.”
According to Vitor Pinheiro, a synthetic biologist at the Rega Institute for Medical Research in Belgium, “The laureates laid out some of the early principles of directed evolution of proteins and inspired many, like me, to look at biology as a flexible canvas that can be shaped to a desired function.”
Directed evolution sparked a revolution because it allowed researchers to mimic the same process that nature used to create the endless diversity of species that fills the world. With his theory of evolution through natural selection, Charles Darwin identified how an iterative process of genetic mutation, selection by the environment, and proliferation by the winners could, over generations, allow organisms to become better at solving complex problems of survival. In the same way, protein chemists can use artificial processes of mutation and selection to coax living cells to find biochemical solutions to problems that would be intractably hard for humans to solve through planning or trial and error.
“Humans have exploited the concept of Darwinian evolution for thousands of years with selective breeding,” said Stefan Lutz, the chairman of the chemistry department at Emory University. “Directed evolution takes this same principle and applies it to the lab.”
By the 1970s, many scientists were dreaming that new genetic engineering technologies might someday be used to create novel proteins with exceptional abilities, such as enzymes that could speed up drug manufacturing. But the hurdle to doing so was huge. “Protein design was an academic exercise. Our computational methods weren’t sophisticated enough to make it practical for designing proteins,” according to Lutz.

During the 1980s, Arnold tried and failed to redesign subtilisin, a bacterial enzyme with diverse commercial applications, so that it could work in solvents other than water. She eventually abandoned that approach in favor of a more evolutionary approach in which she repeatedly induced mutations in bacteria, screened them for the desired traits, and then bred more of them. By 1993, she had succeeded in making the enzyme 256 times more effective than it had been.
“Frances’s insight was to recognize that the most amazing molecules in the world weren’t created by chemists, but rather by the biological world,” said Jesse Bloom, a computational biologist at the Fred Hutchinson Cancer Research Center and a former student of Arnold’s. “Most scientists were accustomed to thinking of evolution in terms of fossils of long-dead creatures, while chemical engineering was something that involved synthetic chemicals in the lab.” The idea of applying evolutionary biology to problems in chemistry and biotechnology, he said, “was a real leap.”
Much of the success of directed evolution in chemistry depended on finding more efficient ways to create and screen for useful forms of new proteins. The foundations of that were laid in 1985, when Smith demonstrated a way to use bacteria and simple viruses called phages to isolate the genes for targeted proteins. He linked genes of interest to the phage virus’s own gene for its protein capsule. With antibodies, he could then isolate any phages that made the protein target.
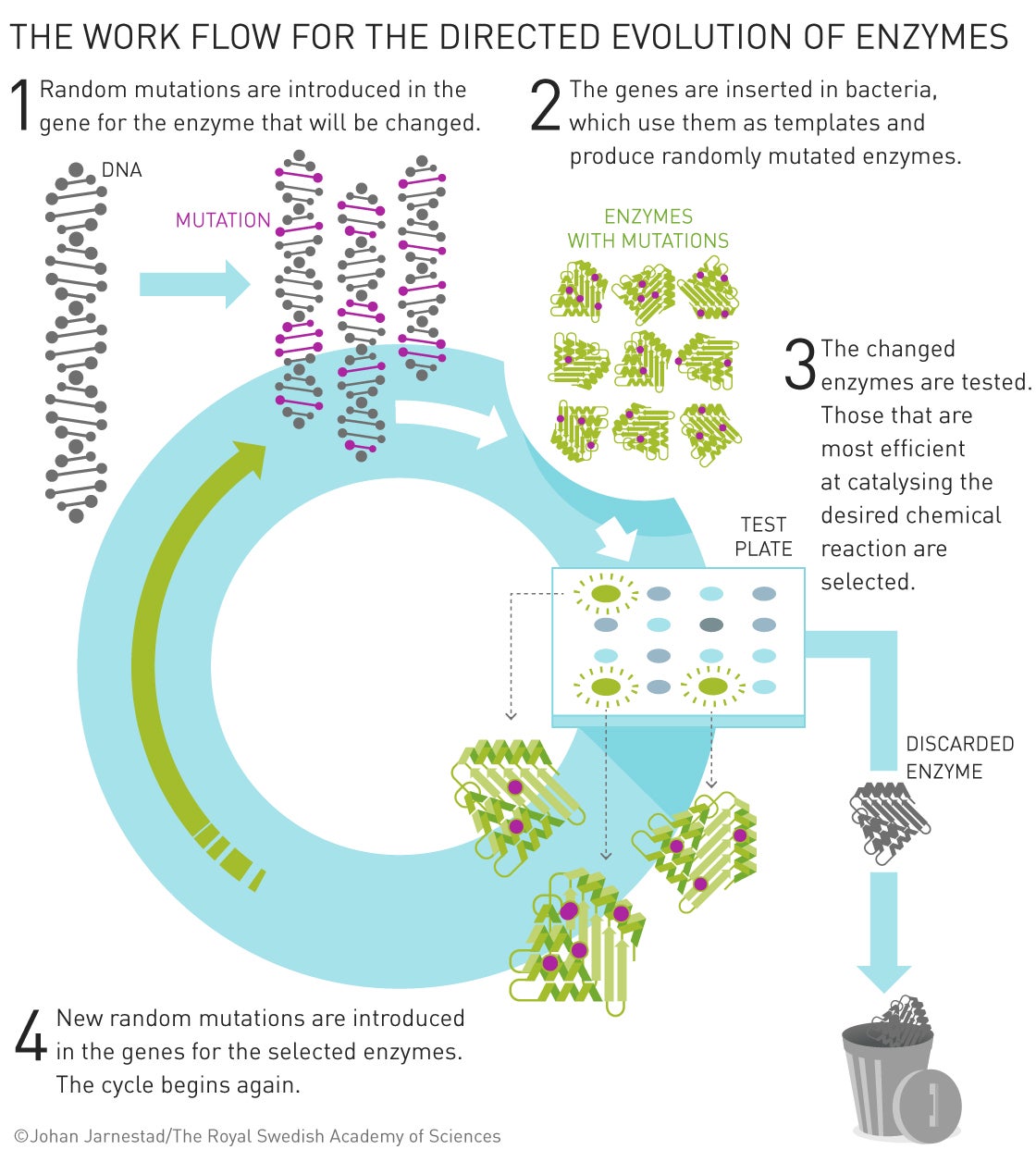
Winter was one of the researchers who subsequently adapted Smith’s techniques to screen for biomolecules with useful activities. In the 1990s he used them to screen for variant forms of antibody proteins that would attach to various biochemical targets. In 2002 a therapeutic antibody based on Winter’s method was approved to treat rheumatoid arthritis and other inflammatory conditions; many other such evolved antibody drugs have followed.
Many more methods for directed evolution have been developed in recent years, but the pioneering contributions of Arnold, Smith, and Winter, along with those of many other scientists who helped to develop the field, are still held in esteem. Arnold still works to further apply directed evolution to pharmaceutical research and renewable energy.
“[Their] pioneering work has enabled and inspired researchers all over the world to harness the power of natural selection for protein engineering,” said Jennifer R. Cochran, a biochemist at Stanford University, via email. Their techniques have become a “backbone of the biotechnology and pharmaceutical industries, further highlighting their societal and commercial impact.”
This article includes contributions from Jordana Cepelewicz and Jonathan Lambert.
Lead image: With genetic engineering techniques and antibodies, researchers can use a process of directed evolution to create useful proteins. Developers of that technique have now been honored with a Nobel Prize in Chemistry. Credit: ustas7777777