The headline on the April 20, 2020, New Yorker magazine profile bestowed a title that has long been unofficial but uncontested: “How Anthony Fauci Became America’s Doctor” it read. Anthony Fauci, MD, has arguably served as “America’s doctor” for decades. He’s been the director of the National Institute of Allergy and Infectious Diseases (NIAID) since 1984. He’s advised seven presidents on countless health issues—domestic and global, emergent and chronic. He oversees the NIAID’s $6 billion budget.
Fauci first came to the public’s attention in the 1980s, as the architect of the nation’s response to the AIDS epidemic. Since early 2020, he has again occupied a national stage, as a trusted voice on COVID-19. He holds the Presidential Medal of Freedom, the National Medal of Science, the Mary Lasker Award for Public Service, and numerous other honors.
This man of the moment is seemingly everywhere: in headlines, on news shows—and, here, sharing his insights on research funding with the Science Philanthropy Alliance. We asked noted science journalist Robert Bazell to interview Fauci as a lead-in to this series of articles on the prequels to humanity’s battle with COVID-19. Bazell was the chief science and health correspondent for NBC News for 38 years, winning five Emmys, a Peabody, and a duPont-Columbia Award. Now an adjunct professor in the Yale Department of Molecular, Cellular, and Developmental Biology, Bazell holds a graduate degree in immunology from the University of California, Berkeley.
The Q&A that follows is based on an interview conducted in January 2021, the transcript of which was condensed and edited for clarity.
Bazell: I’d like to start by asking about the importance of basic science research in the context of COVID-19. One of the things that many people—people not trained in science—are mystified by is the fact that so much foundational knowledge, such as how the cell cycle works, came out of work with yeast or fruit flies. Can you say a few words why such arcane-sounding research—work that to some people might not sound relevant to a global pandemic—matters?
Fauci: That’s very true. In an era of pragmatism, of “what have you done for me lately” thinking, basic science research may not be looked at with a particularly sympathetic eye. But it has been essential to the quick response to COVID-19. For example, a piece of basic research that was key to the development of the vaccines against COVID was the finding that the prefusion form of the coronavirus’s spike protein was infinitely more immunogenic—that is, likely to induce an immune response—than its postfusion form.
Everybody thinks viral molecules are stable, but they’re not. They flip and they flop.
Bazell: I’m sure you remember that when Margaret Heckler famously announced in 1984 that there was going to be an AIDS vaccine in two years, she was widely ridiculed. You and many others went back to the drawing board and learned a lot about HIV, the virus that causes AIDS, and about the correlates of immunity. What role did those advances play in making the world ready to respond to COVID-19?

Fauci: Everything is connected. It became clear to us back in the 1990s that HIV’s monomer spike and certain conformations of its trimer spike were not going to induce broadly neutralizing antibodies. They would only induce homologous antibodies—ones shaped the same as the spikes, that is—and not heterologous antibodies, and this would neutralize the strain, but not broadly.
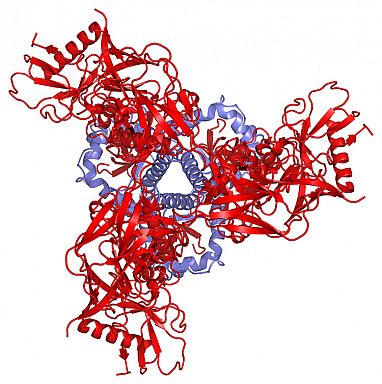
What basic research did, thanks to structural biology and cryo-electron microscopy, was help us figure out how to stabilize the viral envelope so it doesn’t fall apart. Everybody thinks viral molecules are stable, but they’re not. They flip and they flop, so then all of a sudden you’re not inducing neutralizing antibodies, you’re inducing antibodies that are nonneutralizing. A lot of very basic, fundamental work went into showing how you can make mutations that stabilize the conformation of the virus in a way that will be most useful.
Here’s how that happened: Barney Graham—an immunologist who’s interested in RSV, the respiratory syncytial virus—sits down next to structural biologist Peter Kwong and says, “Hey, if I give you this molecule for RSV, can you stabilize this prefusion conformation? Because I know that’s the most immunogenic form, but it keeps falling apart.” What they did is what we did years ago with HIV, which was get back to basics following our failures. They found out that they could take the RSV spike protein and keep it in a stable prefusion conformation—and it was wildly immunogenic, much better than anything that I’ve ever done.
So then along comes COVID, and Barney says, “Wait a minute, we need a vaccine against SARS-CoV-2. Let’s get the spike protein and do the same thing that we were doing with HIV and with RSV and stabilize it in the prefusion form.” Bingo, you’ve got a vaccine that’s 94% to 95% effective. But the reason we were able to get there so quickly with COVID is because of the fundamental, basic research that came out of our efforts on HIV, when I said, “Hey, we’ve got to get back to the basics.” It’s been over 20 years since then, but the same basic idea is what led us so quickly to a vaccine against COVID-19.
Bazell: Billions of dollars in government funding help advance basic research like this. But does or could private philanthropy also play a role?
Fauci: Of course. Right now, in January of 2021, private philanthropists don’t support much basic research. But do they have a role, could they have a role that I think could be really value-added? Absolutely. If private philanthropists were able to fund really high-risk but high-impact fundamental research, that, to me, would be truly value-added. My feeling is that it’s less what have they done than what can they do.
The microbiome is still largely uncharted territory, and it needs to be charted in much more detail.
Bazell: You are often referred to as the nation’s leading infectious disease expert. How much more do we need to know about pathogenesis and immunology in order to respond better to this pandemic and to others that surely lie in our future?
Fauci: The fundamental research that we would welcome much more support for involves, for example, learning more about what we call the prototype pathogen approach. We’d like to be able to say, “What are the commonalities among the different variants of this coronavirus?” So then, when we get a South African mutant and a U.K. mutant, and one of them starts to spread more rapidly and becomes the dominant strain, we can see if there is any aspect of that molecule—when we sequence it as it evolves—that really doesn’t change in the receptor-binding domain or other areas of the spike. If we are able to identify such an area, then we could figure out how to make it very immunogenic and neutralizing, which would allow us to develop a pan-SARS-CoV-2 vaccine—that is, a vaccine that’s effective against all the mutant versions of the virus. There’s a lot of fundamental, basic work that we need to do to get there. And we’re already doing some of it—the NIH is funding a lot of it. But do we need to get more investigators willing to take more risky approaches like that? Of course we do.
Bazell: What you say about taking a risky approach is something that people involved in science policy—including Francis Collins, the director of the National Institutes Health (NIH)—talk about all the time. One of the biggest criticisms of the NIH-funding process is that many of the principal grant mechanisms only fund studies that have a pretty good chance of working. So I’d like to hear more about the role that private philanthropy could play in helping scientists undertake more high-risk, high-reward work.
Fauci: Unfortunately, when it comes to basic research, philanthropy—even big-time philanthropy, like the Gates Foundation—is never going to come anywhere near quantitatively to funding as much as the NIH funds every year with our $42 billion research budget. It’s just not. But what philanthropy can do is something that you brought up yourself, that I talk about a lot, and that Francis Collins does, too, and that is to compensate for one of the weaknesses of the current peer-review funding situation. Although we at the NIH make very effective use of our budget, investigator-initiated research like the work we mostly fund tends not to be very risky. That’s because our peer-review process always seems to start with a question like “What’s your preliminary data?” There’s not much that a scientist with an exciting idea but no data yet can do in the face of such a process. But if private philanthropy could come in and say, “We’re going to put a substantial amount of money into something that is an exciting, risky, potentially high-impact endeavor,” that, I think, is the optimal role for philanthropy.
Bazell: Say a scientist comes to your NIH institute with an idea that prompts other scientists to say, “Boy, that sounds like a long shot. It doesn’t seem like it has much chance of working, but if it did it would be marvelous.” Do you have any thoughts about how scientists with ideas like that can find funding for their work?
Fauci: Yes, I do. One of the strategies that I think is a good model, Bob, is the Howard Hughes Medical Institute model, where they fund people rather than projects. So it would be great if we could fund through private philanthropy investigators who are either young and promising, or who are at a stage where they haven’t yet peaked but are really good, and say to them, “We’re going to fund you for 5 years or 10 years, and you don’t have to worry about wasting any time writing grants. Just give us a couple of very groundbreaking ideas, that are very risky, that you likely would not get traditional grant funding for, and we’re going to support you as you investigate them. We want you to try to fundamentally solve some problem.” If, at the end of one year, you don’t have any results, you don’t need to worry. We’re not going to say, “Uh-oh, your annual report is not good. We’re going to take the money away.” In other words, make risky investments in people who are at the stage of their career where they may inadvertently get sucked into the also-ran track, where they resort to safety, to duplicating or confirming work that’s already been proven because they don’t want to take a chance of not getting funded. To me, that would be a way to get exciting but risky ideas examined.
So I think there is indeed a role for private philanthropy in the process. If you’re a philanthropist and you want to underwrite basic research, looking at high-risk basic research is where you could truly have an effect. You’d need to seek out scientists who have high-risk, high-impact ideas that would not be likely to get funded through the standard NIH peer-review process. That’s what I would recommend.
Bazell: Other than the model of identifying individuals with the right ideas, where should funders put their focus? Are there things that scientists would like to know that you don’t know now, that you can think of on the spot?
Fauci: No, I really can’t do that. That’s both the challenge and the beauty of basic research—you don’t know what you don’t know. It’s only when you discover something new that you realize you didn’t know it before.
Bazell: Can you say how far we are from understanding the immune system, compared to how much you’d like to understand about the immune system?
Fauci: There are certain things about the immune system, particularly related to influenza and imprinting, that we’d love to understand better. In other words, the reason we’ve been so unsuccessful at developing a permanent vaccine against the different strains of influenza, instead of the slightly different annual formulations we now have, is because the immune system seems to have been “imprinted” by earlier strains, and it reverts to responding to that original stimulus. We call it “original antigenic sin.” It means that when the immune system encounters a new variant of a pathogen, it’s apt to respond based on a previous similar pathogen rather than the new one. We still don’t really understand what’s going on there. If that mechanism is something that goes beyond influenza, we’re in a lot of trouble with viruses that mutate, because our immune systems may keep responding to the original imprinting virus rather than to the mutant. That would really be a problem.
Bazell: Perhaps you could think back to the early days of COVID-19, when by definition scientists were looking at a new virus. What do you wish you had known right away that you didn’t know? Is there such a thing?
Fauci: Actually, it was mostly epidemiological questions we were desperately seeking the answers to: How did this new virus spread? How transmissible was it? For example, at first it was thought to have jumped species and not be easily transmissible from human to human. Then we found out it was in fact transmissible from human to human. Then we found it was spectacularly efficient at being transmitted from human to human. Then we found that, “Oh, my god, 50% of transmissions occur from somebody who doesn’t have any symptoms.” We didn’t know that on day one. And that was a game-changer.
Bazell: What about zoonosis—the process of a disease jumping from an animal host to a human host? We know that all humans have microbiomes—a vast array of microbes that live in and on out bodies—and indeed that all creatures have microbiomes. But when the delicate balance in a microbiome goes awry, we get a COVID-like situation. Is there any way we could be better attuned to where the next pathogenic threat might arise?
Fauci: Now you’re talking. The microbiome is definitely an area worthy of inquiry. In fact, some colleagues and I wrote an article about the microbiome that was published just a couple of months ago in the Journal of Infectious Diseases.
We wrote about the need to refocus the research that’s done on the human microbiome; we’ve studied a lot about the microbiome, we’ve described the microbiome, but we really need to know what its impact is. What happens to the microbiome when you give someone a couple of months of antibiotics? What happens when you do this or do that? The microbiome is still largely uncharted territory, and it needs to be charted in much more detail.
Bazell: Is there a connection between understanding the microbiome and understanding the threat of zoonotic diseases?
Fauci: There could be a link there. I think we need to understand how given species either adapt or not to a pathogen. In the case of a virus that jumps from animals to humans, how does it all of a sudden become devastating in humans, when it wasn’t devastating at all in the animal? More specifically, what is it that allows a bat to do fine if it acquires SARS-CoV-2, the virus that causes COVID, but if the same virus gets into a human it creates havoc? I don’t think anybody has a clue about that question.
Bazell: That could be a target for basic research, right?
Fauci: Exactly. You asked earlier, “Is there a role for philanthropy in basic research?” Yes, there is. Are there areas of basic research that I think might be explored with private funding? Yes, there are—high-risk, high-impact, long-term questions. Are there enough unanswered questions that even the $42 billion NIH budget can’t adequately answer them? Yes. The answer to that question is overwhelmingly yes.
Bazell: My last question is how much did basic research have to do with getting you, personally, to the position where you are now—where you’re widely admired and looked to for advice by a significant portion of the world population?
Fauci: I think a lot. I’ve spanned the area between basic research and clinical disease. I decided a very long time ago that I was going to do that, even though it was really difficult. I started studying basic mechanisms of human immunoregulation—very, very fundamental mechanisms—but I’ve also never lost sight of how that understanding is applied. I think that combination was inherent to my being chosen for the position I hold—the fact that I’m, if I might say it myself, a widely respected basic researcher who knows a lot about clinical medicine. That’s what the nation needs in this position.
Bazell: Absolutely.
Lead image credit: Bryce Gladfelter