Anyone who doubts that genes can specify identity might well have arrived from another planet and failed to notice that the humans come in two fundamental variants: male and female. Cultural critics, queer theorists, fashion photographers, and Lady Gaga have reminded us— accurately—that these categories are not as fundamental as they might seem, and that unsettling ambiguities frequently lurk in their borderlands. But it is hard to dispute three essential facts: that males and females are anatomically and physiologically different; that these anatomical and physiological differences are specified by genes; and that these differences, interposed against cultural and social constructions of the self, have a potent influence on specifying our identities as individuals.
That genes have anything to do with the determination of sex, gender, and gender identity is a relatively new idea in our history. The distinction between the three words is relevant to this discussion. By sex, I mean the anatomic and physiological aspects of male versus female bodies. By gender, I am referring to a more complex idea: the psychic, social, and cultural roles that an individual assumes. By gender identity, I mean an individual’s sense of self (as female versus male, as neither, or as something in between).

For millennia, the basis of the anatomical dissimilarities between men and women—the “anatomical dimorphism” of sex—was poorly understood. At the beginning of the third century, Galen, the most influential anatomist in the ancient world, performed elaborate dissections to try to prove that male and female reproductive organs were analogs of each other, with the male organs turned inside out and the female’s turned outside in. The ovaries, Galen argued, were just internalized testicles retained inside the female body because females lacked some “vital heat” that could extrude the organs. “Turn outward the woman’s [organs] and double the man’s, and you will find the same,” he wrote. Galen’s students and followers stretched this analogy, quite literally, to its absurd point, reasoning that the uterus was the scrotum ballooning inward, and that the fallopian tubes were the seminal vesicles blown up and expanded. The theory was memorialized in a medieval verse, an anatomical mnemonic for medical students:
That, tho’ they of different sex may be,
Yet, on the whole, they’re the same as we:
For those that have the strictest searchers been,
Find women are but men turn’d outside in.
But what force was responsible for turning men “inside out,” or women “outside in,” like socks? Centuries before Galen, the Greek philosopher Anaxagoras, writing around the fifth century B.C., claimed that gender, like New York real estate, was determined entirely by location. Like Pythagoras, Anaxagoras believed that the essence of heredity was carried by male sperm, while the female only “shaped” male semen in the womb to produce the fetus. The inheritance of gender also followed this pattern. Semen produced in the right testicle gave rise to male children, while semen produced in the left testicle gave rise to females. The specification of gender continued in the womb, extending the left-right spatial code sparked off during ejaculation. A male fetus was deposited, with exquisite specificity, in the right horn of the uterus. A female, conversely, was nurtured in the left horn.
The Y is the most vulnerable spot in the human genome.
It is easy to laugh Anaxagoras’s theory off as anachronistic and bizarre. Its peculiar insistence on left and right placement—as if gender were determined by some sort of cutlery arrangement—clearly belongs to another era. But the theory was revolutionary for its time, for it made two crucial advances. First, it recognized that the determination of gender was essentially random—and so a random cause (the left or right origin of sperm) would need to be invoked to explain it. And second, it reasoned that once established, the original random act had to be amplified and consolidated to fully engender gender. The developmental plan of the fetus was crucial. Right-sided sperm found its way to the right side of the uterus, where it was further specified into a male fetus. Left-sided sperm was segregated to the left side to make a female child. Sex determination was a chain reaction, set off by a single step but then amplified by the location of the fetus into the full-fledged dimorphism between men and women.
And there, for the most part, sex determination sat, for centuries. Theories abounded, but conceptually they were variants of Anaxagoras’s idea—that sex was determined by an essentially random act, consolidated and amplified by the environment of the egg or fetus. “Sex is not inherited,” one geneticist wrote in 1900. Even the cell biologist Thomas Morgan, who was perhaps the most prominent proponent of the role of genes in development, proposed that sex could not be determined through genes. In 1903, Morgan wrote that sex was likely determined by multiple environmental inputs rather than a single genetic one: “The egg, as far as sex is concerned, appears to be in a sort of balanced state, and the conditions to which it is exposed … may determine which sex it will produce. It may be a futile attempt to try to discover any one influence that has a deciding influence for all kinds of eggs.”

In the winter of 1903, the very year that Morgan had published his casual dismissal of a genetic theory of sex determination, Nettie Stevens, biology Ph.D., performed a study that would transform the field. Stevens was born to a carpenter in Vermont in 1861. She became a schoolteacher, but by the early 1890s, had saved enough money from her teaching jobs to attend Stanford University in California. She chose to attend graduate school in biology in 1900—an unusual choice for a woman in her time—and, even more unusually, chose to perform fieldwork at the zoological station in faraway Naples, where the German embryologist Theodor Boveri had recently isolated chromosomes, in urchin eggs. She learned Italian so that she could speak the lingo of the local fishermen who brought her eggs from the shores. From Boveri, she learned to stain eggs to identify chromosomes—the strange blue-stained filaments that resided in cells.
Boveri had demonstrated that cells with altered chromosomes could not develop normally—and so hereditary instructions for development had to be carried within chromosomes. But could the genetic determinant for sex also be carried by chromosomes? In 1903, Stevens chose a simple organism—the common mealworm—to investigate the correlation between an individual worm’s chromosomal makeup and its sex. When Stevens used Boveri’s chromosome-staining method on male and female worms, the answer leaped out of the microscope: a variation in just one chromosome correlated perfectly with the worm’s sex. Mealworms have 20 chromosomes in all—10 pairs (most animals have paired chromosomes; humans have 23 pairs). Cells from female worms inevitably possessed 10 matched pairs. Cells from male worms, in contrast, had two unpaired chromosomes—a small, nub-like band and a larger chromosome. Stevens suggested that the presence of the small chromosome was sufficient to determine sex. She termed it the sex chromosome. To Stevens, this suggested a simple theory of sex determination. When sperm was created in the male gonad, it was made in two forms—one bearing the nub-like male chromosome, and another bearing the normal size female chromosome—in roughly equal ratios. When sperm bearing the male chromosome—i.e., “male sperm”—fertilized the egg, the embryo was born male. When “female sperm” fertilized an egg, the result was a female embryo.
Stevens’s work was corroborated by that of her close collaborator, the cell biologist Edmund Wilson, who simplified Stevens’s terminology, calling the male chromosome Y, and the female X. In chromosomal terms, male cells were XY, and females were XX. The egg contains a single X chromosome, Wilson reasoned. When a sperm carrying a Y chromosome fertilizes an egg, it results in an XY combination, and maleness is determined. When a sperm carrying an X chromosome meets a female egg, the result is XX, which determines femaleness. Sex was not determined by right or left testicles, but by a similarly random process—by the nature of the genetic payload of the first sperm to reach and fertilize an egg.
The XY system discovered by Stevens and Wilson had an important corollary: If the Y chromosome carried all the information to determine maleness, then that chromosome had to carry genes to make an embryo male. At first, geneticists expected to find dozens of male-determining genes on the Y chromosome: Sex, after all, involves the exacting coordination of multiple anatomical, physiological, and psychological features, and it was hard to imagine that a single gene could be capable of performing such diverse functions all by itself. Yet, careful students of genetics knew that the Y chromosome was an inhospitable place for genes. Unlike any other chromosome, the Y is “unpaired”—i.e., it has no sister chromosome and no duplicate copy, leaving every gene on the chromosome to fend for itself. A mutation in any other chromosome can be repaired by copying the intact gene from the other chromosome. But a Y chromosome gene cannot be fixed, repaired, or recopied from another chromosome; it has no backup or guide (there is, however, a unique internal system to repair genes in the Y chromosome). When the Y chromosome is assailed by mutations, it lacks a mechanism to recover information. The Y is thus pockmarked with the potshots and scars of history. It is the most vulnerable spot in the human genome.
As a consequence of this constant genetic bombardment, the human Y chromosome began to jettison information millions of years ago. Genes that were truly valuable for survival were likely shuffled to other parts of the genome where they could be stored securely; genes with limited value were made obsolete, retired, or replaced; only the most essential genes were retained (some were duplicated in the Y chromosome itself—but even this strategy does not solve the problem completely). As information was lost, the Y chromosome itself shrank—whittled down piece by piece by the mirthless cycle of mutation and gene loss. That the Y chromosome is the smallest of all chromosomes is not a coincidence: it is largely a victim of planned obsolescence (in 2014, scientists discovered that a few extremely important genes may be permanently lodged in the Y).
In genetic terms, this suggests a peculiar paradox. Sex, one of the most complex of human traits, is unlikely to be encoded by multiple genes. Rather, a single gene, buried rather precariously in the Y chromosome, must be the master regulator of maleness. Male readers of that last paragraph should take notice: We barely made it.
In the early 1980s, a young geneticist in London named Peter Goodfellow began to hunt for the sex-determining gene on the Y chromosome. A die-hard soccer enthusiast—scruffy, bone-thin, taut, with an unmistakable East Anglian drawl and a “punk meets new romantic” dress sense—Goodfellow intended to use a gene-mapping method pioneered by David Botstein and Ron Davis a few years before to narrow the search to a small region of the Y chromosome. But how could a “normal” gene be mapped without the existence of a variant phenotype, or an associated disease? By 1989, cystic fibrosis and Huntington’s disease genes had been mapped to their chromosomal locations by tracking the link between the disease-causing gene and signposts along the genome. In both cases, affected siblings carrying the gene also carried the signpost, while unaffected siblings did not. But where might Goodfellow find a human family with a variant gender—a third sex—that was genetically transmitted, and carried by some siblings but not others?
By flicking a single genetic switch, Goodfellow had switched an organism’s sex.
In fact, such humans existed—although identifying them was a much more complicated task than anticipated. In 1955, Gerald Swyer, an English endocrinologist investigating female infertility, had discovered a rare syndrome that made humans biologically female but chromosomally male. “Women” born with “Swyer syndrome” were anatomically and physiologically female throughout childhood, but did not achieve female sexual maturity in early adulthood. When their cells were examined, geneticists discovered that these “women” had XY chromosomes in all their cells. Every cell was chromosomally male—yet the person built from these cells was anatomically, physiologically, and psychologically female. A “woman” with Swyer syndrome had been born with the male chromosomal pattern (i.e., XY chromosomes) in all of her cells, but had somehow failed to signal “maleness” to her body.
The most likely scenario behind Swyer syndrome was that the master-regulatory gene that specifies maleness had been inactivated by a mutation, leading to femaleness. At the Massachusetts Institute of Technology, a team led by the geneticist David Page had used such sex-reversed women to map the male-determinant gene to a relatively narrow region of the Y chromosome. The next step was the most laborious—the gene-by-gene sifting to find the correct candidate among the dozens of genes in that general location. Goodfellow was making slow, steady progress when he received devastating news. In the summer of 1987, he learned that Page had landed on the male-determinant gene. Page called the gene ZFY, for its presence in the Y chromosome.
Initially, ZFY seemed like the perfect candidate: It was located in the right region of the Y chromosome, and its DNA sequence suggested that it could act as a master switch for dozens of other genes. But when Goodfellow looked carefully, the shoe wouldn’t fit: When ZFY was sequenced in women with Swyer syndrome, it was completely normal. There was no mutation that would explain the disruption of the male signal in these women. With ZFY disqualified, Goodfellow returned to his search. The gene for maleness had to be in the region identified by Page’s team: They must have come close, but just missed it. In 1989, rooting about close to the ZFY gene, Goodfellow found another promising candidate—a small, nondescript, tightly packed, intronless gene called SRY. Right at the onset, it seemed like the perfect candidate. The normal SRY protein was abundantly expressed in the testes, as one might expect for a sex-determination gene. Other animals, including marsupials, also carried variants of the gene on their Y chromosomes—and thus only males inherited the gene. The most striking proof that SRY was the correct gene came from the analysis of human cohorts: The gene was indisputably mutated in females with Swyer syndrome, and nonmutated in their unaffected siblings.
But Goodfellow had one last experiment to clinch the case—the most dramatic of his proofs. If the SRY gene was the singular determinant of “maleness,” what if he forcibly activated the gene in female animals? Would females be forced to turn into males? When Goodfellow inserted an extra copy of the SRY gene into female mice, their offspring were born with XX chromosomes in every cell (i.e., genetically female), as expected. Yet the mice developed as anatomically male—including growing a penis and testicles, mounting females, and performing every behavior characteristic of male mice. By flicking a single genetic switch, Goodfellow had switched an organism’s sex—creating Swyer syndrome in reverse.
Is all of sex just one gene, then? Almost. Women with Swyer syndrome have male chromosomes in every cell in the body—but with the maleness-determining gene inactivated by a mutation, the Y chromosome is literally emasculated (not in a pejorative but in a purely biological sense). The presence of the Y chromosome in the cells of women with Swyer syndrome does disrupt some aspects of the anatomical development of females. In particular, breasts do not form properly, and ovarian function is abnormal, resulting in low levels of estrogen. But these women feel absolutely no disjunction in their physiology. Most aspects of female anatomy are formed perfectly normally: The vulva and vagina are intact, and a urinary outlet is attached to them with textbook fidelity. Astonishingly, even the gender identity of women with Swyer syndrome is unambiguous: just one gene flicked off and they “become” women. Although estrogen is undoubtedly required to enable the development of secondary sexual characteristics and reinforce some anatomical aspects of femininity in adults, women with Swyer syndrome are typically never confused about gender or gender identity. As one woman wrote, “I definitely identify with female gender roles. I’ve always considered myself 100 percent female. I played on a boy’s soccer team for a while—I have a twin brother; we look nothing alike—but I was definitely a girl on a boy’s team. I didn’t fit in well: I suggested that we name our team ‘the butterflies.’ ”
Women with Swyer syndrome are not “women trapped in men’s bodies.” They are women trapped in women’s bodies that are chromosomally male (except for just one gene). A mutation in that single gene, SRY, creates a (largely) female body—and, more crucially, a wholly female self. It is as artless, as plain, as binary, as leaning over the nightstand and turning a switch on or off.

If genes determine sexual anatomy so unilaterally, then how do genes affect gender identity? On the morning of May 4, 2004, David Reimer, a 38-year-old man in Winnipeg, drove into the parking lot of a grocery store and killed himself with a sawed-off shotgun. Born in 1965 as Bruce Reimer—chromosomally, and genetically, male—David had been the victim of a ghoulish attempt at circumcision by an inept surgeon, resulting in a severely damaged penis in early infancy. Reconstructive surgery was impossible, and so Bruce’s parents had rushed him to see John Money, a psychologist at Johns Hopkins University, known internationally for his interest in gender and sexual behavior. Money evaluated the child and, as part of an experiment, asked Bruce’s parents to have their son castrated and raise him as a girl. Desperate to give their son a “normal” life, his parents capitulated. They changed his name to Brenda.
Money’s experiment on David Reimer—for which he never asked or received permission from the university or hospital—was an attempt to test a theory widely fashionable in academic circles in the ’60s. The notion that gender identity was not innate and was crafted through social performance and cultural mimicry (“you are who you act; nurture can overcome nature”) was in its full prime in that era—and Money was among its most ardent and most vocal proponents. Casting himself as the Henry Higgins of sexual transformation, Money advocated “sexual reassignment,” the reorientation of sexual identity through behavioral and hormonal therapy—a decades-long process invented by his team at Johns Hopkins that allowed his experimental subjects to emerge with their identities sanguinely switched. Based on Money’s advice, “Brenda” was dressed and treated as a girl. Her hair was grown long. She was given female dolls and a sewing machine. Her teachers and friends were never informed about the switch. Brenda had an identical twin—a boy named Brian—who was brought up as a male child. As part of the study, Brenda and Brian visited Money’s clinic in Baltimore at frequent intervals throughout their childhood. As preadolescence approached, Money prescribed estrogen supplements to feminize Brenda. The surgical construction of an artificial vagina was scheduled to complete her anatomical transformation into a woman. Money published a steady stream of highly cited papers touting the extraordinary success of the sexual reassignment. Brenda was adjusting to her new identity with perfect equanimity, he proposed. Her twin, Brian, was a “rough and tumble” boy, while Brenda was an “active little girl.” Brenda would ease into womanhood with scarcely any hurdles, Money declared. “Gender identity is sufficiently incompletely differentiated at birth to permit successful assignment of a genetic male as a girl.”
It is now clear that genes are vastly more influential than virtually any other force in shaping sex identity and gender identity.
In reality, nothing could have been further from the truth. At age 4, Brenda took scissors and shredded the pink and white dresses she had been forced to wear. She lapsed into fits of fury when told to walk or talk like a girl. Pinioned to an identity that she found evidently false and discordant, she was anxious, depressed, confused, anguished, and often frankly enraged. In her school reports, Brenda was described as “tomboyish” and “dominant,” with “abundant physical energy.” She refused to play with dolls or other girls, preferring her brother’s toys (the only time she played with her sewing machine was when she sneaked a screwdriver out of her father’s toolbox and took the machine meticulously apart, screw by screw). Perhaps most confoundingly to her young classmates, Brenda went to the girl’s bathroom dutifully—but then preferred to urinate with her legs spread wide, standing up.
After 14 years, Brenda was told about the circumstances surrounding her birth, and she brought the grotesque charade to an end. She refused the vaginal operation. She stopped the estrogen pills, underwent a bilateral mastectomy to excise her breast tissue, and began injecting testosterone to revert back to male. She—he—changed her name to David. He married a woman in 1990, but the relationship was tormented from the start. Bruce/Brenda/David—the boy who became a girl who became a man—continued to ricochet between devastating bouts of anxiety, anger, denial, and depression. He lost his job. The marriage failed. In 2004, shortly after a bitter altercation with his wife, David killed himself.
David Reimer’s case was not unique. In the 1970s and 1980s, several other cases of sexual reassignment—the attempted conversion of chromosomally male children into females through psychological and social conditioning—were described, each troubled and troubling in its own right. In some cases, the gender dysphoria was not as acute as David’s— but the wo/men often suffered haunting pangs of anxiety, anger, dysphoria, and disorientation well into adulthood. In one particularly revealing case, a woman—called C—came to see a psychiatrist in Rochester, Minnesota. Dressed in a frilly, floral blouse and a rough cowhide jacket—“my leather-and-lace look,” as she described it—C had no problems with some aspects of her duality, yet had trouble reconciling her “sense of herself as fundamentally female.” Born and raised as a girl in the 1940s, C recalled being a tomboy in school. She had never thought of herself as physically male, but had always felt a kinship with men (“I feel like I have the brain of a man”). She married a man in her 20s and lived with him—until a chance ménage à trois involving a woman kindled her fantasies about women. Her husband married the other woman, and C left him and entered a series of lesbian relationships. She oscillated between periods of equanimity and depression. She joined a church and discovered a nurturing spiritual community—except for a pastor who railed against her homosexuality and recommended therapy to “convert” her.
At 48, goaded by guilt and fear, she finally sought psychiatric assistance. During the medical examination, her cells were sent for chromosomal analysis, and she was found to have XY chromosomes in her cells. Genetically speaking, C was male. She later discovered that s/he had been born with ambiguous, underdeveloped genitals, although chromosomally male. Her mother had consented to reconstructive surgery to transform her into a female. Sexual reassignment had begun when she was 6 months old, and she had been given hormones at puberty on the pretext of curing a “hormonal imbalance.” Throughout her childhood and adolescence, C did not have the faintest spasm of doubt about her gender. C’s case illustrates the importance of thinking carefully about the link between gender and genetics. Unlike David Reimer, C was not confused about the performance of gender roles: She wore female clothes in public, maintained a heterosexual marriage (for a while, at least), and acted within the range of cultural and social norms to pass as female for 48 years. Yet despite her guilt about her sexuality, crucial aspects of her identity—kinship, fantasy, desire, and erotic drive—remained fastened to maleness. C had been able to learn many of the essential features of her acquired gender through social performance and mimesis, but she couldn’t unlearn the psychosexual drives of her genetic self.
In 2005, a team of researchers at Columbia University validated these case reports in a longitudinal study of “genetic males”—i.e., children born with XY chromosomes—who had been assigned to female gender at birth, typically because of the inadequate anatomical development of their genitals. Some of the cases were not as anguished as David Reimer’s or C’s—but an overwhelming number of males assigned to female gender roles reported experiencing moderate to severe gender dysphoria during childhood. Many had suffered anxiety, depression, and confusion. Many had voluntarily changed genders back to male upon adolescence and adulthood. Most notably, when “genetic males” born with ambiguous genitals were brought up as boys, not girls, not a single case of gender dysphoria or gender change in adulthood was reported.
These case reports finally put to rest the assumption, still unshakably prevalent in some circles, that gender identity can be created or programmed entirely, or even substantially, by training, suggestion, behavioral enforcement, social performance, or cultural interventions. It is now clear that genes are vastly more influential than virtually any other force in shaping sex identity and gender identity—although in limited circumstances a few attributes of gender can be learned through cultural, social, and hormonal reprogramming. Since even hormones are ultimately “genetic”—i.e., the direct or indirect products of genes—then the capacity to reprogram gender using purely behavioral therapy and cultural reinforcement begins to tip into the realm of impossibility. Indeed, the growing consensus in medicine is that, aside from exceedingly rare exceptions, children should be assigned to their chromosomal (i.e., genetic) sex regardless of anatomical variations and differences—with the option of switching, if desired, later in life. As of this writing, none of these children have opted to switch from their gene-assigned sexes.
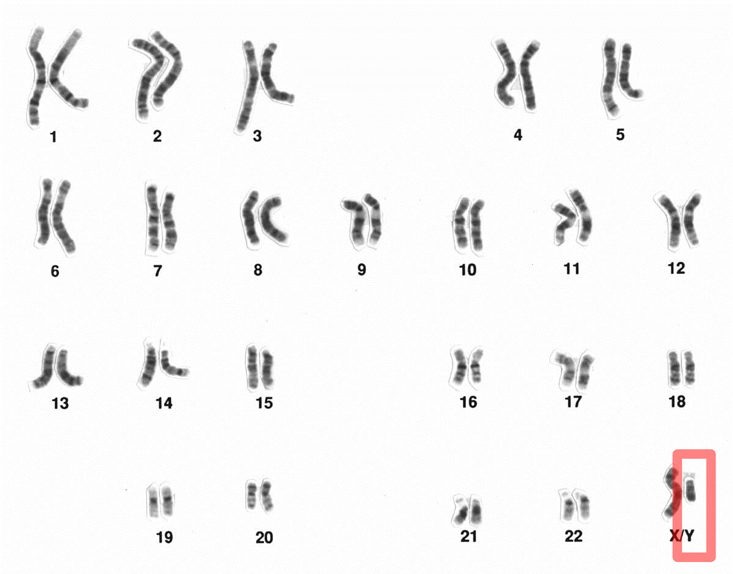
How can we reconcile this idea—of a single genetic switch that dominates one of the most profound dichotomies in human identity—with the fact that human gender identity in the real world appears in a continuous spectrum? Virtually every culture has recognized that gender does not exist in discrete half-moons of black and white, but in a thousand shades of gray. Even Otto Weininger, the Austrian philosopher now famous for his misogyny, conceded, “Is it really the case that all women and men are marked off sharply from each other … ? There are transitional forms between the metals and nonmetals; between chemical combinations and simple mixtures, between animals and plants, between phanerogams and cryptogams, and between mammals and birds. … The improbability may henceforth be taken for granted of finding in Nature a sharp cleavage between all that is masculine on the one side and all that is feminine on the other.”
In genetic terms, though, there is no contradiction: Master switches and hierarchical organizations of genes are perfectly compatible with continuous curves of behavior, identity, and physiology. The SRY gene indubitably controls sex determination in an on/off manner. Turn SRY on, and an animal becomes anatomically and physiologically male. Turn it off, and the animal becomes anatomically and physiologically female.
But to enable more profound aspects of gender determination and gender identity, SRY must act on dozens of targets—turning them on and off, activating some genes and repressing others, like a relay race that moves a baton from hand to hand. These genes, in turn, integrate inputs from the self and the environment—from hormones, behaviors, exposures, social performance, cultural role-playing, and memory—to engender gender. What we call gender, then, is an elaborate genetic and developmental cascade, with SRY at the tip of the hierarchy, and modifiers, integrators, instigators, and interpreters below. This geno-developmental cascade specifies gender identity. Genes are like single lines in a recipe that specifies gender. The SRY gene is the first line in the recipe: “Start with four cups of flour.” If you fail to start with the flour, you will certainly not bake anything close to a cake. But infinite variations fan out of that first line—from the crusty baguette of a French bakery to the eggy mooncakes of Chinatown.
The existence of a transgender identity provides powerful evidence for this geno-developmental cascade. In an anatomical and physiological sense, sex identity is quite binary: Just one gene governs sex identity, resulting in the striking anatomical and physiological dimorphism that we observe between males and females. But gender and gender identity are far from binary. Imagine a gene—call it TGY—that determines how the brain responds to SRY (or some other male hormone or signal). One child might inherit a TGY gene variant that is highly resistant to the action of SRY on the brain, resulting in a body that is anatomically male, but a brain that does not read or interpret that male signal. Such a brain might recognize itself as psychologically female; it might consider itself neither male or female, or imagine itself belonging to a third gender altogether.
These men (or women) have something akin to a Swyer syndrome of identity: Their chromosomal and anatomical gender is male (or female), but their chromosomal/anatomical state does not generate a synonymous signal in their brains. In rats, notably, such a syndrome can be caused by changing a single gene in the brains of female embryos or exposing embryos to a drug that blocks the signaling of “femaleness” to the brain. Female mice engineered with this altered gene or treated with this drug have all the anatomical and physiological features of femaleness, but perform the activities associated with male mice, including mounting females: these animals might be anatomically female, but they are behaviorally male.
The hierarchical organization of this genetic cascade illustrates a crucial principle about the link between genes and environments in general. The perennial debate rages on: nature or nurture, genes or environment? The battle has gone on for so long, and with such animosity, that both sides have capitulated. Identity, we are now told, is determined by nature and nurture, genes and environment, intrinsic and extrinsic inputs. But this too is nonsense—an armistice between fools. If genes that govern gender identity are hierarchically organized—starting with SRY on top and then fanning out into thousands of rivulets of information below—then whether nature predominates or nurture is not absolute, but depends quite acutely on the level of organization one chooses to examine.
At the top of the cascade, nature works forcefully and unilaterally. Up top, gender is quite simple—just one master gene flicking on and off. If we learned to toggle that switch—by genetic means or with a drug—we could control the production of men or women, and they would emerge with male versus female identity (and even large parts of anatomy) quite intact. At the bottom of the network, in contrast, a purely genetic view fails to perform; it does not provide a particularly sophisticated understanding of gender or its identity. Here, in the estuarine plains of crisscrossing information, history, society, and culture collide and intersect with genetics, like tides. Some waves cancel each other, while others reinforce each other. No force is particularly strong—but their combined effect produces the unique and rippled landscape that we call an individual’s identity.
From: The Gene: an Intimate History by Siddhartha Mukherjee. Copyright © 2016 by Siddhartha Mukherjee, M.D. Reprinted by permission of Scribner, a Division of Simon & Schuster, Inc.
Lead photo collage credit: Cultura RM Exclusive/Luc Beziat / Dimitri Otis / Getty Images
This article was originally published in Nautilus Magazine on December 29, 2016.